Biodegradable nanocomposites incorporating carbon nanofibers (CNFs) have gained significant traction due to their environmentally friendly nature. The use of functionalized CNFs enhances the mechanical, thermal, and electrical properties of nanocomposites. The ultimate properties and biodegradation rate of these nanocomposites are significantly influenced by the type and structure of the CNFs dispersed within the biodegradable polymer matrix. Nanocomposites were prepared by blending 0.2% w/w of the functionalized butyl, and dodecyl CNFs in cellulose acetate polymer matrix. The study sought to establish the effect of the butyl, and dodecyl moieties on the degradation rate of biodegradable cellulose acetate. Scanning Electron Microscopy (SEM) and Energy Dispersive Spectroscopy (EDS) were used to determine the dispersion of CNFs within the polymer matrix and the surface characteristics of the resulting nanocomposites. Respirometry (CO2 emission) and gravimetry (change in mass) techniques were used to determine the biodegradation rate of the nanocomposites. The study found out that incorporation of functionalized CNFs into the biodegradable polymer matrix had an impact on the biodegradation rates of the formed nanocomposites. From the cumulative amounts of CO2 evolved during the respirometry and cumulative weight lost during the test period, the nanocomposites had a reduced rate of degradation compared to the reference blank. This could be attributed to an increase in polymer crystallinity caused by the addition of the alky moieties that increased the adherence of the CNFs to the polymer matrix. Individual alky functionalized nanocomposite also had different rates of degradation with the butyl nanocomposite degrading much faster than the dodecyl, respectively. Overall, the results indicated a slight increase in the time required for the nanocomposite to degrade to less than 1% of the original sample as compared to the reference blank. The study and its findings have generated new scientific knowledge that could be relevant in the fabrication biodegradable nanocomposites based on a diverse range of other polymeric and nonpolymeric matrices and importantly approximately how long the fibers can be in the environment after their useful life.
| Published in | International Journal of Environmental Chemistry (Volume 9, Issue 2) |
| DOI | 10.11648/j.ijec.20250902.14 |
| Page(s) | 62-71 |
| Creative Commons |
This is an Open Access article, distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium or format, provided the original work is properly cited. |
| Copyright |
Copyright © The Author(s), 2025. Published by Science Publishing Group |
Carbon Nanofibers, Nanocomposites, Biodegradation
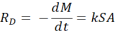 (2)
(2) 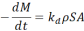 (3)
(3) 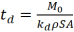 =(5)
=(5) Sample | 14 days | 28 days | 42 days | 56 days | 70 days |
|---|---|---|---|---|---|
Control set up | 21.56 | 30.58 | 18.96 | 23.32 | 15.18 |
Dodecyl CNC | 24.21 | 36.20 | 28.12 | 33.61 | 15.41 |
Butyl CNC | 35.22 | 35.24 | 39.61 | 42.42 | 15.48 |
Reference blank | 39.63 | 37.42 | 46.21 | 38.42 | 22.63 |
Sample | 14 days | 28 days | 42 days | 56 days | 70 days |
|---|---|---|---|---|---|
Dodecyl CNC | 2.65 | 5.62 | 9.16 | 10.29 | 0.23 |
Butyl CNC | 13.66 | 4.66 | 20.65 | 19.1 | 0.3 |
Reference blank | 18.07 | 6.84 | 27.25 | 15.1 | 7.45 |
Sample | 14 days | 28 days | 42 days | 56 days | 70 days |
|---|---|---|---|---|---|
Dodecyl CNC | 2.65 | 8.27 | 17.48 | 27.72 | 27.95 |
Butyl CNC | 13.66 | 18.32 | 38.97 | 58.07 | 58.37 |
Reference blank | 18.07 | 24.91 | 52.16 | 67.26 | 74.71 |
Sample | Mo | Ms-14 days | Ms -28 days | Ms -42 days | Ms -56 days | Ms -70 days |
|---|---|---|---|---|---|---|
Dodecyl CNC | 0.4995 | 0.4441 | 0.4331 | 0.3911 | 0.3566 | 0.3282 |
Butyl CNC | 0.5009 | 0.4540 | 0.4442 | 0.3831 | 0.3496 | 0.3216 |
Ref. Blank | 0.5090 | 0.4765 | 0.4321 | 0.3418 | 0.3291 | 0.3099 |
Sample | 14 days | 28 days | 42 days | 56 days | 70 days |
|---|---|---|---|---|---|
Dodecyl CNC | 88.9% | 86.7% | 78.3% | 71.4% | 65.7% |
Butyl CNC | 90.6% | 88.7% | 76.5%) | 69.8%) | 64.2% |
Reference Blank | 93.8 | 84.9% | 67.2% | 64.7% | 60.9% |
Sample | Mi | Ms | Δ mass (g) | RD = ΔM /t |
|---|---|---|---|---|
Dodecyl CNC | 0.4995 | 0.3284 | 0.1711 | 0.00244 |
Butyl CNC | 0.5009 | 0.3215 | 0.1794 | 0.00256 |
Reference | 0.5090 | 0.3101 | 0.1989 | 0.00284 |
Sample | Mo (g) | RD (g/day | SA (cm2) | ρ (g/cm3) | Kd (cm/day) | td (day) |
|---|---|---|---|---|---|---|
Dodecyl CNC | 0.4995 | 0.00244 | 4.186 | 1.964 | 0.00029679 | 204.71 |
Butyl CNC | 0.5009 | 0.00256 | 4.186 | 1.641 | 0.000372677 | 195.66 |
Reference | 0.509 | 0.00284 | 4.186 | 1.41 | 0.000481172 | 179.23 |
CA | Cellulose Acetate |
CNF | Carbon Nanofibers |
CTAB | Cetyltrimethylammonium Bromide |
EDS | Energy Dispersive Spectroscopy |
PTFE | Polytetrafluoroethylene |
PMMA | Poly Methylmethacrylate |
SA | Surface Area |
SEM | Scanning Electron Microscopy |
SSDR | Specific Surface Degradation Rate |
| [1] | Biswal, T., BadJena, S. K., & Pradhan, D. (2020). Synthesis of polymer composite materials and their biomedical applications. Materials Today: Proceedings, 30, 305-315. |
| [2] | Goodwin, D. G., Boyer, I., Devahif, T., Gao, C., Frank, B. P., Lu, X., Kuwama, L., Gordon, T. B., Wang, J., Ranville, J. F., Bouwer, E. J., & Fairbrother, D. H. (2018). Biodegradation of Carbon Nanotube/Polymer Nanocomposites using a Monoculture. Environmental Science & Technology, 52(1), 40-51. |
| [3] | Kikku Fukushima et al. (2009), Biodegradation of poly (lactic acid) and its nanocomposites, Polymer Degradation and Stability 94 1646-1655, |
| [4] | Hoshino, A., Tsuji, M., Ito, M., Momochi, M., Mizutani, A., Takakuwa, K., Higo, S., Sawada, H., & Uematsu, S. (2003). Study of the Aerobic Biodegradability of Plastic Materials under Controlled Compost. In E. Chiellini & R. Solaro (Eds.), Biodegradable Polymers and Plastics (pp. 47-54). Springer US. |
| [5] | Roy, S. B., Shit, S. C., Sengupta, R. A., & Shukla, P. R. (2015). Biodegradability studies of bio-composites of polypropylene reinforced by potato starch. Int J Innov Res Sci Eng Technol, 4(3), 1120-30. |
| [6] | Sanchez-Garcia, M. D., Lagaron, J. M., & Hoa, S. V. (2010). Effect of addition of carbon nanofibers and carbon nanotubes on properties of thermoplastic biopolymers. Composites Science and Technology, 70(7), 1095-1105. |
| [7] | Chamas, A., Moon, H., Zheng, J., Qiu, Y., Tabassum, T., Jang, J. H., Abu-Omar, M., Scott, S. L., & Suh, S. (2020). Degradation Rates of Plastics in the Environment. ACS Sustainable Chemistry & Engineering, 8(9), 3494-3511. |
| [8] | Doppalapudi, S., Jain, A., Khan, W. and Domb, A. J. (2014), Biodegradable polymers-an overview†. Polym. Adv. Technol., 25: 427-435. |
| [9] | Bonilla, J.; Sobral, P. (2020) Disintegrability under composting conditions of films based on gelatin, chitosan and/or sodium caseinate containing boldo-of-Chile leafs extract. Int. J. Biol. Macromol., 151, 178-185, |
| [10] | Karlsson, T. M., Hassellöv, M., & Jakubowicz, I. (2018). Influence of Thermo oxidative degradation on the in-situ fate of polyethylene in temperate coastal waters. Marine Pollution Bulletin, 135, 187-194. |
| [11] | Fatimah Alshehrei, (2017): “Biodegradation of Synthetic and Natural Plastic by Microorganisms.” Journal of Applied & Environmental Microbiology, vol. 5, no. 1 8-19. |
| [12] | Agamuthu, P.; Faizura, P. N (2005). Biodegradability of Degradable Plastic Waste. Waste Manage. Res., 23(2), 95-100, |
| [13] | Bernstein, R.; Derzon, D. K.; Gillen, K. T. (2005). Nylon 6.6 Accelerated Aging Studies: Thermal-Oxidative Degradation and Its Interaction with Hydrolysis. Polym. Degrad. Stab., 88(3), 480-488, |
| [14] | Zaghdoudi, M., Kömmling, A., Jaunich, M., & Wolff, D. (2020). Erroneous or Arrhenius: A degradation rate-based model for EPDM during homogeneous ageing. Polymers, 12(9), 2152 |
| [15] | Selene Chinaglia et al. 2018: Polymer Degradation and Stability 147 237-244 |
| [16] | Othman, S. H.; Ronzi, N. D. A.; Shapi’i, R. A.; Dun, M.; Ariffin, S. H.; Mohammed, M. A. P. (2023). Biodegradability of Starch Nanocomposite Films Containing Different Concentrations of Chitosan Nanoparticles in Compost and Planting Soils. Coatings, 13, 777. |
| [17] | Tamada, J. A.; Langer, R. (1993). Erosion Kinetics of Hydrolytically Degradable Polymers. Proc. Natl. Acad. Sci. U. S. A., 90(2), 552-556. |
| [18] | Sharon, C.; Sharon, M. (2017). Studies on Biodegradation of Polyethylene Terephthalate: A Synthetic Polymer. J. Microbiol. Biotechnol. Res., 2(2), 248-257, |
| [19] | Kausar, A., & Bocchetta, P. (2022). Poly (methyl methacrylate) nanocomposite foams reinforced with carbon and inorganic nanoparticles State-of-the-art. Journal of Composites Science, 6(5), 129, |
| [20] | Liang, F., Sadana, A. K., Peera, A., Chattopadhyay, J., Gu, Z., Hauge, R. H., & Billups, W. E. (2004). A Convenient Route to Functionalized Carbon Nanotubes. Nano Letters, 4(7), 1257-1260. |
| [21] | Vatansever, A., Inan, T. Y., Dogan, H., & Sirkecioglu, A. (2015). Synthesis of poly butyl acrylate‐co‐methyl methacrylate)/montmorillonite waterborne nanocomposite via semi-batch emulsion polymerization. Journal of Applied Polymer Science, 132(32), app. 42373. |
| [22] | Murali, A., Ramkumar, S. C., Haridharan, N., Mohana, S., Samanta, D., & Jaisankar, S. N. (2020). Multifunctional properties of acetaminophen immobilized polymer nanohybrid composites. SN Applied Sciences, 2(7), 1313. |
| [23] | Eubeler, J. P., Zok, S., Bernhard, M., & Knepper, T. P. (2009). Environmental biodegradation of synthetic polymers I. Test methodologies and procedures. TrAC Trends in Analytical Chemistry, 28(9), 1057-1072. |
| [24] | Cindradewi, A. W., Bandi, R., Park, C.-W., Park, J.-S., Lee, E.-A., Kim, J.-K., Kwon, G.-J., Han, S.-Y., & Lee, S.-H. (2021). Preparation and characterization of cellulose acetate film reinforced with cellulose nanofibril. Polymers, 13(17), 2990, |
| [25] | Singh, N. K.; Purkayastha, B. D.; Roy, J. K.; Banik, R. M.; Yashpal, M.; Singh, G.; Malik, S.; Maiti, P. (2009) Nanoparticle-induced controlled biodegradation and its mechanism in poly (ε-caprolactone) ACS Appl. Mater. Interfaces, 2(1), 69-81, |
| [26] | Wang, S., Song, C., Chen, G., Guo, T., Liu, J., Zhang, B., & Takeuchi, S. (2005). Characteristics and biodegradation properties of poly (3-hydroxybutyrate-co-3-hydroxyvalerate)/organophilic montmorillonite (PHBV/OMMT) nanocomposite. Polymer Degradation and Stability, 87(1), 69-76. |
APA Style
Mukhebi, S. S., Owour, J., Otieno, G., Aluoch, A. O., Andala, D. M. (2025). Comparative Biodegradation Studies of Butyl and Dodecyl - Functionalized Carbon Nanofibers Dispersed in Cellulose Acetate Polymer Nanocomposites. International Journal of Environmental Chemistry, 9(2), 62-71. https://doi.org/10.11648/j.ijec.20250902.14
ACS Style
Mukhebi, S. S.; Owour, J.; Otieno, G.; Aluoch, A. O.; Andala, D. M. Comparative Biodegradation Studies of Butyl and Dodecyl - Functionalized Carbon Nanofibers Dispersed in Cellulose Acetate Polymer Nanocomposites. Int. J. Environ. Chem. 2025, 9(2), 62-71. doi: 10.11648/j.ijec.20250902.14
@article{10.11648/j.ijec.20250902.14,
author = {Situma Stephen Mukhebi and James Owour and Geoffrey Otieno and Austin Ochieng Aluoch and Dickson Mubera Andala},
title = {Comparative Biodegradation Studies of Butyl and Dodecyl - Functionalized Carbon Nanofibers Dispersed in Cellulose Acetate Polymer Nanocomposites
},
journal = {International Journal of Environmental Chemistry},
volume = {9},
number = {2},
pages = {62-71},
doi = {10.11648/j.ijec.20250902.14},
url = {https://doi.org/10.11648/j.ijec.20250902.14},
eprint = {https://article.sciencepublishinggroup.com/pdf/10.11648.j.ijec.20250902.14},
abstract = {Biodegradable nanocomposites incorporating carbon nanofibers (CNFs) have gained significant traction due to their environmentally friendly nature. The use of functionalized CNFs enhances the mechanical, thermal, and electrical properties of nanocomposites. The ultimate properties and biodegradation rate of these nanocomposites are significantly influenced by the type and structure of the CNFs dispersed within the biodegradable polymer matrix. Nanocomposites were prepared by blending 0.2% w/w of the functionalized butyl, and dodecyl CNFs in cellulose acetate polymer matrix. The study sought to establish the effect of the butyl, and dodecyl moieties on the degradation rate of biodegradable cellulose acetate. Scanning Electron Microscopy (SEM) and Energy Dispersive Spectroscopy (EDS) were used to determine the dispersion of CNFs within the polymer matrix and the surface characteristics of the resulting nanocomposites. Respirometry (CO2 emission) and gravimetry (change in mass) techniques were used to determine the biodegradation rate of the nanocomposites. The study found out that incorporation of functionalized CNFs into the biodegradable polymer matrix had an impact on the biodegradation rates of the formed nanocomposites. From the cumulative amounts of CO2 evolved during the respirometry and cumulative weight lost during the test period, the nanocomposites had a reduced rate of degradation compared to the reference blank. This could be attributed to an increase in polymer crystallinity caused by the addition of the alky moieties that increased the adherence of the CNFs to the polymer matrix. Individual alky functionalized nanocomposite also had different rates of degradation with the butyl nanocomposite degrading much faster than the dodecyl, respectively. Overall, the results indicated a slight increase in the time required for the nanocomposite to degrade to less than 1% of the original sample as compared to the reference blank. The study and its findings have generated new scientific knowledge that could be relevant in the fabrication biodegradable nanocomposites based on a diverse range of other polymeric and nonpolymeric matrices and importantly approximately how long the fibers can be in the environment after their useful life.},
year = {2025}
}
TY - JOUR T1 - Comparative Biodegradation Studies of Butyl and Dodecyl - Functionalized Carbon Nanofibers Dispersed in Cellulose Acetate Polymer Nanocomposites AU - Situma Stephen Mukhebi AU - James Owour AU - Geoffrey Otieno AU - Austin Ochieng Aluoch AU - Dickson Mubera Andala Y1 - 2025/08/27 PY - 2025 N1 - https://doi.org/10.11648/j.ijec.20250902.14 DO - 10.11648/j.ijec.20250902.14 T2 - International Journal of Environmental Chemistry JF - International Journal of Environmental Chemistry JO - International Journal of Environmental Chemistry SP - 62 EP - 71 PB - Science Publishing Group SN - 2640-1460 UR - https://doi.org/10.11648/j.ijec.20250902.14 AB - Biodegradable nanocomposites incorporating carbon nanofibers (CNFs) have gained significant traction due to their environmentally friendly nature. The use of functionalized CNFs enhances the mechanical, thermal, and electrical properties of nanocomposites. The ultimate properties and biodegradation rate of these nanocomposites are significantly influenced by the type and structure of the CNFs dispersed within the biodegradable polymer matrix. Nanocomposites were prepared by blending 0.2% w/w of the functionalized butyl, and dodecyl CNFs in cellulose acetate polymer matrix. The study sought to establish the effect of the butyl, and dodecyl moieties on the degradation rate of biodegradable cellulose acetate. Scanning Electron Microscopy (SEM) and Energy Dispersive Spectroscopy (EDS) were used to determine the dispersion of CNFs within the polymer matrix and the surface characteristics of the resulting nanocomposites. Respirometry (CO2 emission) and gravimetry (change in mass) techniques were used to determine the biodegradation rate of the nanocomposites. The study found out that incorporation of functionalized CNFs into the biodegradable polymer matrix had an impact on the biodegradation rates of the formed nanocomposites. From the cumulative amounts of CO2 evolved during the respirometry and cumulative weight lost during the test period, the nanocomposites had a reduced rate of degradation compared to the reference blank. This could be attributed to an increase in polymer crystallinity caused by the addition of the alky moieties that increased the adherence of the CNFs to the polymer matrix. Individual alky functionalized nanocomposite also had different rates of degradation with the butyl nanocomposite degrading much faster than the dodecyl, respectively. Overall, the results indicated a slight increase in the time required for the nanocomposite to degrade to less than 1% of the original sample as compared to the reference blank. The study and its findings have generated new scientific knowledge that could be relevant in the fabrication biodegradable nanocomposites based on a diverse range of other polymeric and nonpolymeric matrices and importantly approximately how long the fibers can be in the environment after their useful life. VL - 9 IS - 2 ER -